- Blog
- Black ops 2 esp mod
- Constructor game torrent
- Colin mcrae rally 3 cheats keygen
- Oziexplorer per mac
- Telecharger pes 4 demo
- Five degrees of separation
- Slave maker 3 apk
- Valentina studio preferences
- Logitech c525 web cam
- Vray for c4d 3-25
- Kinsey scale test vistriai
- Harry potter film release date
- Teisco del rey guitar sunburst
- Revman 5 collaboration
- Trados studio 2017 auto propagation
- Zinstall winwin cracked
- Lil boosie superbad return of mr wipe me down
- High eq meaning
- Ganesh shadakshari mantra
- Leaving neverland debunked
- The 100 season 6 episode 1 leaked
- Apa format generator
- Ipadian premium crackeado
- Dae jo yeong 7 eng sub dramacool
- The experiment 2010 -srt
- Radiant max phone case
- Engraving trophies
- Plants vs zombies garden warfare 2 characters scientist
- 1990 crx 4 speed
- Manu punjabi
- Gujarati songs falguni pathak
- Score high ranch rush 2
- Alsife ternary diagram
- I surrender celine dion itunes
- Super singer 6 vijay tv
- Rurouni kenshin kyoto inferno misao
- Clinical neuroanatomy made ridiculously simple free pdf
- Xojo reviews
- Ashrae psychrometric chart registration how to
- Federal e i number

#Revman 5 collaboration trial
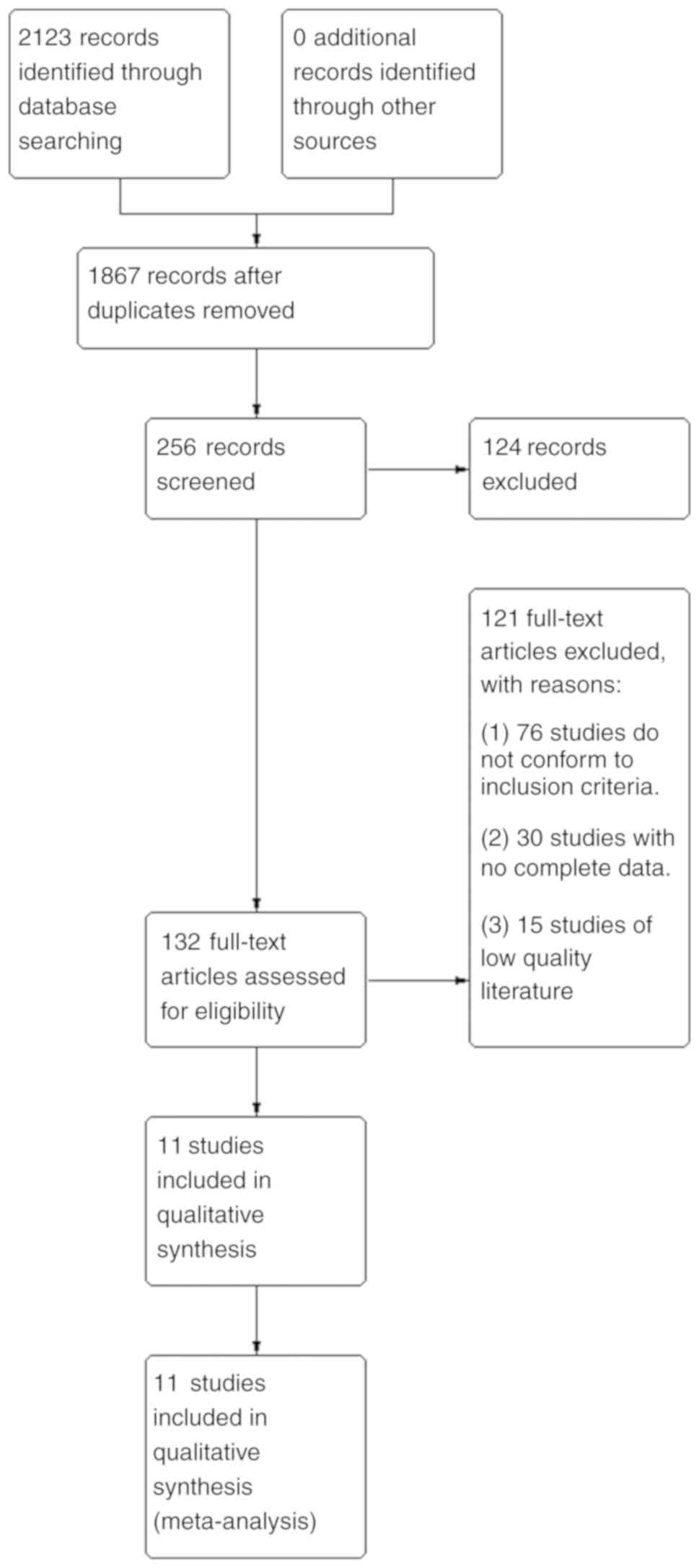
The items on the list were divided into seven areas: generation of the allocation sequence concealment of the allocation sequence blinding attrition and exclusions other generic sources of bias biases specific to the trial design (such as crossover or cluster randomised trials) and biases that might be specific to a clinical specialty. Before the meeting, JPTH and DGA compiled an extensive list of potential sources of bias in clinical trials. In May 2005, 16 statisticians, epidemiologists, and review authors attended a three day meeting to develop the new tool. In this paper we describe the collaboration’s new risk of bias assessment tool, and the process by which it was developed and evaluated. 8 In 2005 the Cochrane Collaboration’s methods groups embarked on a new strategy for assessing the quality of randomised trials. 4 5 6 7 Until recently, Cochrane reviews used a variety of these tools, mainly checklists. Many tools for assessing the quality of randomised trials are available, including scales (which score the trials) and checklists (which assess trials without producing a score). The notion of study “quality” is not well defined but relates to the extent to which its design, conduct, analysis, and presentation were appropriate to answer its research question. To obtain reliable conclusions, review authors must carefully consider the potential limitations of the included studies. Systematic reviews aim to collate and synthesise all studies that meet prespecified eligibility criteria 3 using methods that attempt to minimise bias. 2 However, it is usually impossible to know the extent to which biases have affected the results of a particular trial. 1Ĭausal inferences from randomised trials can, however, be undermined by flaws in design, conduct, analyses, and reporting, leading to underestimation or overestimation of the true intervention effect (bias). Differences in outcomes of interest between the different groups can then in principle be ascribed to the causal effect of the intervention. Provided that there are enough participants, randomisation should ensure that participants in the intervention and comparison groups are similar with respect to both known and unknown prognostic factors. Randomised trials, and systematic reviews of such trials, provide the most reliable evidence about the effects of healthcare interventions. The Cochrane Collaboration’s tool for assessing risk of bias aims to make the process clearer and more accurate
.+Version+5.2+The+Cochrane+Collaboration%2C.jpg)
